Water Management is a critical issue for Planet Earth.
Did you know less than 3% of the Earth’s water is fresh drinking water?
71% of the planet’s surface is covered by water. Yet, the world suffers from water scarcity. Limited supply coupled with population growth is worsening the problem.
Still, reports show that about 40% of the world’s population live within 100 km of the ocean. Is Desalination therefore the answer to our water scarcity?

What is Desalination?
Desalination is any process that remove excess salt and other minerals from water. Its purpose is to make fresh water that is suitable for humans to drink, or to use for domestic and industrial purposes.
It is, for example, very helpful for reducing salt levels in water that cause corrosion of metals.

Desalination plants use one of two main technologies:
- Membrane technologies: This is a distillation process. It can be ‘multi-stage flash’, ‘multi-effect’, ‘vapor-compression’, or ‘solar’ distillation.
- Thermal technologies: This includes ‘electrodialysis’ and ‘electrodialysis reversal’ and ‘reverse osmosis’.
The most common method is reverse osmosis. This process works as follows:
Desalination Steps
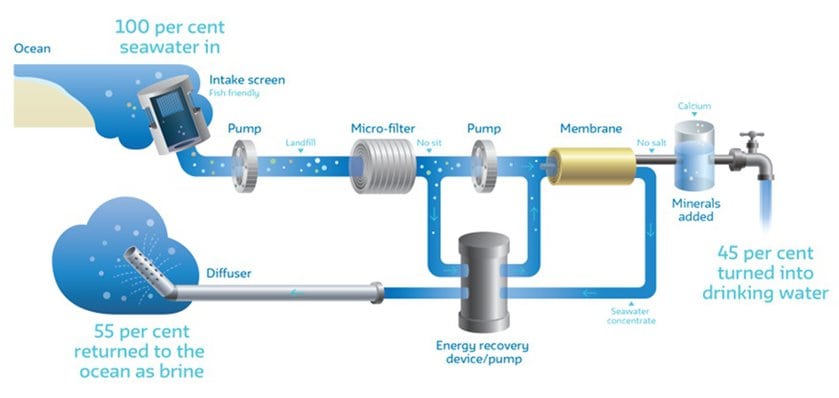
Step One: Obtaining Seawater
The first step is to get the source – seawater. Usually, the plant pipes in seawater from an ocean depth of at least 5 meters. At this point, the water is devoid of fish but may contain small substances or particles such as microbes, particles, and salt (mostly sodium chloride, but also other compounds).
Step two: Pre-treatment
In pre-treatment, the water goes through filters to take out any particles, biological matter, and other impurities. Pre-treatment can use sand-based filters or ultrafiltration membranes. Then, another filter called a cartridge filter completes this step.
Step three: Reverse Osmosis
A series of very thin membranes at very high pressure removes salt from the water. It passes through incredibly tiny, roughly atom-sized holes. In this way, water molecules pass through, while salt stays on the other side. The hydrostatic pressure used in reverse osmosis is around 700 meters (which is extremely high!)
Step four: The Parting of Ways
The end result is not dry salt and pure water. It’s very pure water and very salty water. So, what happens to that very salty water? The plant pumps it directly back into the ocean, at a speed of four meters per second.
Step five: De-purifying the Pure Product
Believe it or not, completely pure water is actually quite dangerous to drink. So, the water needs a final process. Promineralisation makes pure water drinkable. An injection of carbon dioxide and liquid lime adds hardness. Finally, the water leaves the plant, ready for use.
Water Management with Desalination
Pros
- Desalination provides potable water as a substitute source of freshwater
The process of seawater desalination uses the ocean’s unlimited water source. It therefore protects freshwater resources and its habitat.
- Safe water quality without harming any living things
Desalination produces water that is safe and of high quality. It eases water scarcity and drought in certain areas. Also, unlike surface water sources, it does not contain harmful chemicals or pollutants.
- Uses tried-and-tested technology
Desalination is a tried and tested process. Many industries and applications have used the process over many years. Results prove it is an effective method for producing safe and reliable drinking water. In addition, it may also increase our awareness of the need to protect our oceans.
Cons
- Desalination plants are very costly to build and operate
Depending on the location, a desalination plant costs between $300 million to $2.9 billion to build. The operational cost is also very high. About one third to one half of that is due to energy costs. So, desalination plants are very sensitive to the price of power.
On a positive note, desalination plants are often located in sunny areas. Therefore, the use of solar power can reduce costs. In addition, the price of building plants has declined as technologies have improved.
- They have a harmful impact on the environment
The biggest issue is byproducts. The disposal of salt, known as brine, needs to be responsible. Otherwise, salinity and lowered oxygen levels will damage ocean waters. These pose a threat to local flora and fauna. In addition, other chemical by-products can be harmful in high concentrations. These include chlorine, hydrochloric acid, and anti-scalents.
However, studies show that there are economic opportunities with brine. It can be used for commercial salt, metal recovery, and in fish production systems.
Conclusion
Desalination has proven to be an excellent resource for water management. It offers a powerful solution to a world facing increased scarcity from climate change. However it is also very expensive and has a lot of ongoing costs. Nevertheless, understanding the limits of desalination and managing the associated risks can make it a very safe and valuable addition to water policy.
To learn more about water management and the development of sustainable resources visit the THRIVE blog. We invite you to join THRIVE and be part of the change. Let’s build a ‘thrivable’ world where human societies and the environment not only survive, but thrive, well into the future.